|
3/24/2024 0 Comments Bohr's model of the atom  
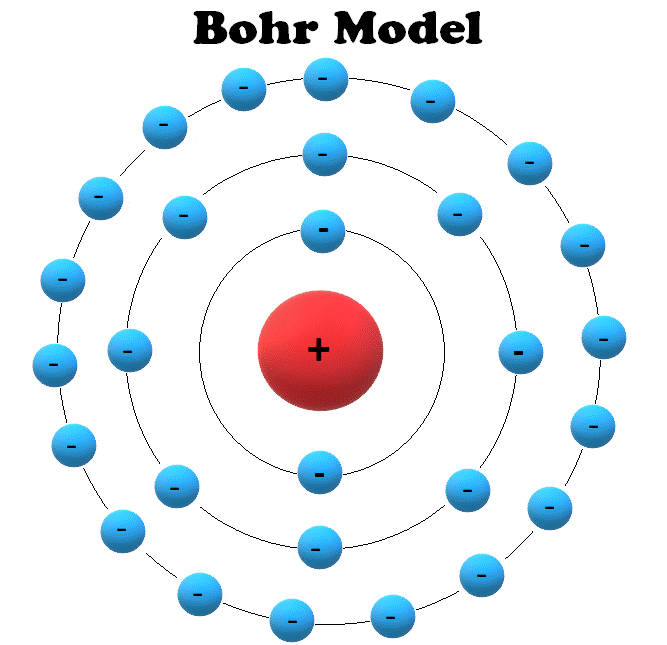
These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells. The figure includes a diagram representing the relative energy levels of the quantum numbers of the hydrogen. How did scientists figure out the structure of atoms without looking at them Try out different models by shooting light at the atom. Because a hydrogen atom with its one electron in this orbit has the lowest possible energy, this is the ground state (the most stable arrangement of electrons for an element or a compound), the most stable arrangement for a hydrogen atom. The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus (Figure 1). Figure 6.2.2: The horizontal lines show the relative energy of orbits in the Bohr model of the hydrogen atom, and the vertical arrows depict the energy of photons absorbed (left) or emitted (right) as electrons move between these orbits. Bohrs Model of an Atom was proposed in 1913 as a modification of the prevailing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
